Translate this page into:
Pulse oximetry as a tool in the physiologic validation of oxygen systems for helicopters
Abstract
Aero Medical Training Center, Hindon was tasked to find holistic and practical solutions to the problems of oxygen systems in Cheetah, Mi-17 and Mi-17-1 V helicopters, presently flying in the high altitude terrain. Additional task was to find highest possible degree of commonalisation in different helicopters. A study was undertaken to physiologically validate various oxygen systems already in use and to select an ideal system for each helicopter. Altitude chamber based and in flight trials were carried out using the oxygen systems in use and their ergonomically suitable modifications. The three basic regulators tested were FA-06, KP-58 and the Ulmer regulators. A total of 4 sets of chamber trials and 6 sets of flight trials were conducted. SPO2 was recorded using Nonin Pulse Oximeter (Model 8600 with finger probe) to assess the adequacy of oxygen delivery. The preliminary studies and the first flight trial revealed the need to change the user interface (mask / helmet combination) in ali the helicopters. It also highlighted the need to evaluate the KP-58 regulator physiologically for Mi-17 and a need to replace fixed dilution regulator with an automatic one, in Cheetah. Chamber trials were done to test the newly developed TAN mask with all the regulators being tested. In addition, the demand diluter functions of KP-58 and Ulmer regulators were confirmed. Modification of Cheetah oxygen bottle with KP-58 regulator also functioned satisfactorily. Based on these trials the oxygen system for each type of helicopter on physiologic, ergonomic and commonalisation related considerations were recommended. Pulse oximetry was found to be a highly useful tool for field research in hypoxia and oxygen system related research.
Keywords
Pulse oximetry
Oxygen system
Physiologic validation
Aero Medical Training Center, Hindon was tasked to find holistic and practical solutions to the problems of oxygen systems in the Cheetah, Mi-17 and Mi-17-lV helicopters, presently flying in the high altitude terrain. The guidelines given were to achieve maximum physiological adequacy to match the operational capabilities of the respective helicopters and to achieve highest possible degree of commonalisation.
Preliminary review and assessment highlighted the lacunae in the various oxygen systems (Table 1). The pilot interface, viz. oxygen mask in all the helicopters and integrated helmet in Cheetah, was inadequate and needed to be addressed on priority. A new mask, common for use in all the helicopters and an integrated Cheetah helmet were conceived and prototypes evaluated by 1 AMTC in Jul 2001[1].
The next issue was to provide automatic, demand-diluter, man-mounted regulator that would replace the regulators in Mi-17 and Cheetah. The oxygen system provided in Mi-17 IV (Table 1) had such a regulator, but it had not been physiologically evaluated in India till date. It was premised that, if found physiologically adequate, this combination could be readily introduced in Mi-17 as it is. In Cheetah, preliminary aircraft trials revealed that this system could not be readily incorporated on account of the large size of the oxygen bottle incompatible with the cockpit dimensions. To tackle this issue, a two-pronged approach was planned:
Combining Cheetah original oxygen bottle with KP-75 reducer and combining two such bottles with KP-58 regulator and TAN mask (Fig 1).
Simultaneous procurement of Ulmer™ (French) regulator and it’s testing in combination with Cheetah original bottle with built in reducer and TAN mask. The three oxygen regulators that were tested during the trials are shown in Fig 1.
Materials and Methods
Four sets of Altitude Chamber trials and six in-flight trials were conducted. Helicopter pilots flying Cheetah and Mi-17 helicopters were used as subjects. Altitude chamber trials were conducted at the existing Decompression Chamber at 1 AMTC. Flight trials were conducted in Cheetah helicopters belonging to helicopter units at Hindan, Sarsawa and Leh. Nonin (model 8600)
| Cheetah (EROS) | MI-17 (KKO-PC) | MI-17 IV (KKO-PC2) | |
|---|---|---|---|
| Bottle Pressure | 150 bar | 30 Ks/cm2 | 210Kg/cm2 |
| Reducer | EROS | KP-21 | KP-75A |
| Mask | Modified ABEU | KM15N | KM16H |
| Helmet | AB EU with inner | Leather inner | Leather inner |
| Microphone | Mask intergrated | Ext Boom microphone | Larvneophone |

- Three regulators tested as part of the trials
-
FA-06 Manual fixed dilution (0, 50,100%) regulator (Presently in use in Cheetah)
-
KP-58 Automatic Demand Diluter Regulator (Presently in use in Mi-17 IV)
-
Ulmer™ Automatic Demand diluter Regulator (Tested for Army Aviation in 1998)
FA-06 Manual fixed dilution (0, 50,100%) regulator (Presently in use in Cheetah)
KP-58 Automatic Demand Diluter Regulator (Presently in use in Mi-17 IV)
Ulmer™ Automatic Demand diluter Regulator (Tested for Army Aviation in 1998)
Pulse Oximeter, with an adult finger probe, placed on the index finger of subjects, was used to measure the percentage saturation of haemoglobin (SPO2) (Fig 2). The newly developed helicopter oxygen mask and integrated helmet, being inducted in the IAF, were used in all the chamber and flight trials.

- Nonin™ PULSE OXIMETER (model, 8600) with Finger Probe
General Guidelines: The details of the guidelines followed in the altitude chamber and flight trials is given in subsequent paragraphs :
(a) Altitude Chamber Trials
(i) All the chamber trials were conducted in the morning hours after ensuring that the aircrew had adequate rest and breakfast. The aircrew were asked to avoid alcohol on the day prior. Ear clearance run to rule out eustachian tube dysfunction was given before the actual trials. For chamber trials, in-built Mk 17-E oxygen regulator was used as positive control.
(ii) The following two types of trials were given in the chamber:
(aa) Physiologic Adequacy Ron. The chamber was raised to maximum altitude of 21,000 ft at ascent / descent rates of 3,000 ft/min. SPO2 was noted, both during ascent and descent, at every 2,000 ft till 20,000 ft and at 21,000 ft. In addition, any subjective problems such as breathing resistance, mask leakage, etc., reported by the pilot, were studied within the chamber. A recorded SPO0 value of <65% was used as safety cut. off to abandon the trial for that modification and the subject using that specific modification was then asked to change over to built-in Mk-17 E regulator of the chamber. The chamber profile for the Physiologic Adequacy Run is shown schematically in Figure 3(A).
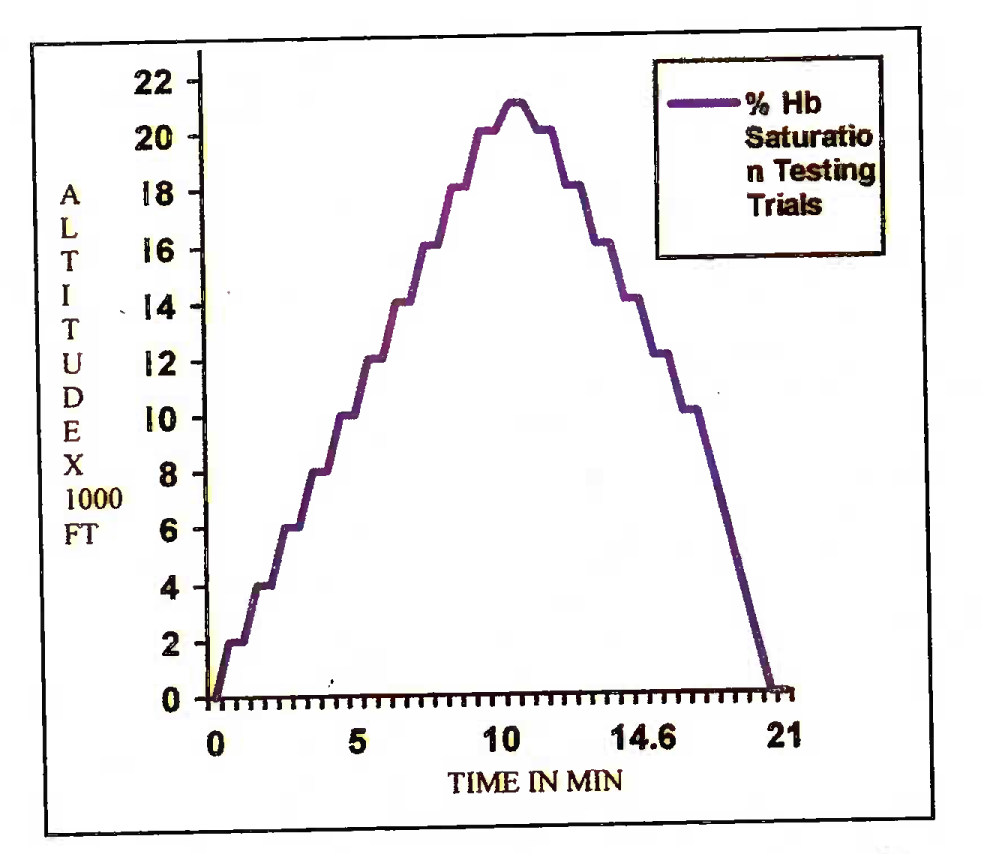
- Physiologic adequacy run profile (CT 1)
- Notes:
- * Rate of chamber ascent = 3,000 ft/min
- * Horizontal lines represent the time of recording pulse oximetry reading
- * Altitude of recording: Every 2,000 ft up to 20,000 ft and then at 21,000 ft
- * No recording between 10,000 ft to ground level in descent
Notes:
* Rate of chamber ascent = 3,000 ft/min
* Horizontal lines represent the time of recording pulse oximetry reading
* Altitude of recording: Every 2,000 ft up to 20,000 ft and then at 21,000 ft
* No recording between 10,000 ft to ground level in descent
(ab) Endurance Run. The chamber was raised to 15,000 ft and the subject asked to breathe normally. The under-test oxygen systems were switched on at 10,000 ft and the first SPO2 recording was done at 10,000 ft. Subsequent SPO2 recordings were done at 15,000 ft at every 10 minutes of exposure as a safety check. The oxygen cylinder, charged to 110 bar pressure (≈110 Kg/ cm2), was weighed before the trial. The end-points for cessation of the trial was drop in the cylinder pressure to 10 bar (≈10 Kg/cm2). The chamber was brought to ground level and the cylinder was weighed again at the end of the the trial. Total oxygen consumed was noted along with the system endurance, defined as the time from switching on the system at 10,000 ft to drop in cylinder pressure to 10 bar. A total of 6 subjects were tested on each of the systems and average endurance was calculated. The chamber profile for Endurance Test is shown schematically in Fig 3(B).
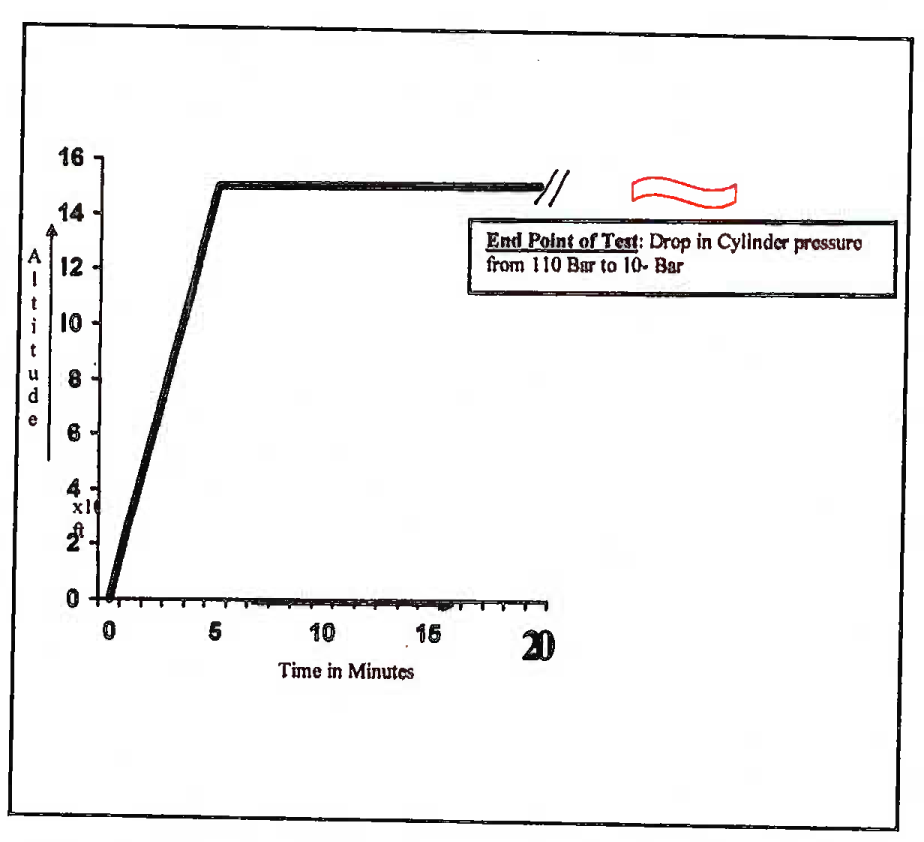
- Altitude chamber flight profile: Oxygen endurance testing
(b) Flight Trials
(i) One aviation medicine specialist (authors) along with a technical officer, were present on board in all the trials. These two passengers were used as additional subjects for testing additional configurations of the oxygen system under testing. The oxygen system configuration, found satisfactory in the chamber trials, was only taken up for flight trials. One of the aircrew was maintained on the presently used oxygen system (Table 1), since this system is flight certified for Cheetah at present. The other aircrew was given the configuration under check. System endurance in minutes was noted along with the pressure and weight difference in the oxygen cylinder before and after the flight. A total of 6 trials each were done and the average endurance of the trials on each system calculated. A photograph of the oxygen system modification under test, mounted in the Cheetah cockpit during flight trials is shown in Fig 4.
(ii) The following two types of recording profiles were done in-flight:

- One of the modifications mounted in Cheetah cockpit
(aa) Flight Trial 1 (FT1). SPO2 was recorded on ground at Leh in eight aircrew. The subject aircrew were seated as passengers in Cheetah helicopter (total of 5 sorties) and flown to altitude of 18,000 ft. At 18,000 ft, the SPO2 was measured again. The subjects were then asked to don the existing oxygen system mask with regulator setting at 50% and SPO2 was measured. Then the subjects were asked to convert to 100% setting and SPO2 was measured again.
(ab) Subsequent Flight Trials After takeoff the aircraft climbed to max flight-planned altitude (which depended on local weather, route etc). SPO2 recording was commenced at 10,000 ft MSL and was recorded at every 10 minutes of flight in all the subjects.
Results and Discussion
The various altitude chamber and flight based pulse oximetry trials conducted are summarized in Table 2. The table also summarizes the aims of testing and the details of the various combinations tested.
| Trial Type | Systems Tested | Aim of Trials |
|---|---|---|
| Flight Trial 1 (FT-1) | Cheetah O2 Bottle + FA-06 + modified ABEU Mask | Preliminary Assessment |
| Chamber Test 1 (CTI-1) | (a) Cheetah O2 Bottle + FA-06+EROS Mask (b) Cheetah O2 Bottle + FA-06-t-Modified ABEU Mask (c) Cheetah O2 Bottle + FA-06+Customized TAN Mask (d) Mi 17 lVO2Bottle+KP-75/KP-58+ Customized TAN Mask |
(a) To Confirm the shortcomings of (i) EROS mask & FA-06 Combination (ii) Modified ABEU mask and FA-06 combination (b) To assess improvements of Customized TAN masks and FA-06 combination (c) To establish efficacy of Mi-17IV system with customized TAN mask. |
| Chamber Test 2 (CT-2) | Single Cheetah O2 Bottle modified to fit KP-75+KP-58+ Customized TAN Mask | (a) To assessphysiologic efficacy of commonalised system in single bottle configuration (b) To compare the system endurance for FA-06+ABEU combination with KP 75 + KP-58 + customized TAN mask combination. |
| Flight Trial 2 (FT-2) | (a ) Cheetah O2 Bottle + FA-06 + Modified ABEU Mask. (b) Single Cheetah O2 Bottle modified to fit KP-75+KP 58 + Customized TAN Mask |
” |
| Chamber Test 3 (CT-3) | Two sets of Cheetah O2 Bottle modified to fit KP-75+KP-58+Customized TAN Mask interconnected with the junction box developed by 3 BRD | To assess physiologic efficacy of commonalised system in double bottle configuration-tested on a passenger subject |
| Flight Trial 3 (FT -3) | ” | To assess physiologic efficacy of commonalised system in double configuration aircrew subject (System installed in-situ with one bottle behind the pilot and the other behind the instrument panel) |
| Flight Trial 4 (FT-4) | ” | ” (System installed in-situ with both bottles behind the pilot seat on either side) |
| Chamber Trial 4 (CT-4) | Cheetah O2 Bottle +Ulmer + Customized Tan mask | (a) To assess the physiologic adequacy of Ulmer regulator in combination with TAN mask |
| Flight Trial 5 (FT-5) | (a) Flight confirmation of CT4 (b) Comparison of double-bottle prototype with Ulmer prototype |
|
| Fhght Trial 6 (FT - 6) | ” | ” |
Evaluation of Trial Results
(a) Flight trial 1 (FT 1)
(i) The SPO2 values on ground (Leh airfield base altitude of 10,682 ft MSL) in the 8 pilots showed a mean of 93.4 + 2.6% (range 91 to 98%). At 18,000 ft MSL with normal air breathing, 50% oxygen enrichment and 100% oxygen the SPO2 values were 82.5 + 2.7% (range 77 to 86%), 88.6 + 2.97% (range of 86 to 93%) and 93.1 ± 1.4% (range of 90 to 94%) respectively (Table - 3).
| Subject No | On Ground (10,682 ft MSL) (in %) | Without Oxygen | At 18,000 ft MSL (in %) At 50% Regulator Setting | At 100% Regulator Setting |
|---|---|---|---|---|
| 1 | 91 | 82 | 87 | 93 |
| 2 | 93 | 77 | 89 | 94 |
| 3 | 93 | 86 | 89 | 93 |
| 4 | 91 | 83 | 86 | 90 |
| 5 | 96 | 85 | 86 | 93 |
| 6 | 91 | 83 | 86 | 94 |
| 7 | 94 | 83 | 93 | 94 |
| 8 | 98 | 81 | 93 | 94 |
| Mean | 93.4 | 82.5 | 88.6 | 93.1 |
| SD | ±2.6 | ±2.7 | ±2.97 | ±1.4 |
(ii) This first trial was done to assess the extent and the nature of the problems of the in-use Cheetah oxygen system (Table -1). Lower than expected levels of SPO2 in some of the pilots on ground (10,682 ft MSL) and variability in the values· in different subjects is due to the differences in their acclimatization status. At 18,000 ft, 38 to 48% enriched oxygen is recommended to maintain > 90% SPO2 [2]. A less than 90% SPO2, even on breathing 50% oxygen enriched breathing mixture, clearly indicated a problem with the delivery system (mask leakage and high breathing resistance). Qualitatively, it was also observed that saturation values varied, depending on the depth of breathing,which further confirmed the high breathing resistance. This fact had been independently confirmed in an earlier study at LAM [3]. On breathing 100% oxygen, SPO2 values increased to above 90%. However, breathing 100% oxygen caused serious depletion of the oxygen on board, thereby reducing the system endurance (vis-a-vis the aircraft endurance) and hence was not an acceptable solution. Thus, data presented in this table helped understanding the actual oxygenation derived by the pilots through fixed 50% and 100% settings of the FA-06 regulator. It clearly indicated that there was a need to improve the delivery system (mask) to achieve the goal of improving the oxygen system for Cheetah in addition to addressing the problems of manual and fixed dilution problems of the FA-06 regulator.
(b) Chamber trial 1 (CT 1)
(i) The SPO2 values, recorded in the specified different combinations of FA-06 regulator with EROS, ABEU and TAN masks as a function of altitude are shown in Figure 5(A) and tabulated in Table 4. In EROS and ABEU combinations, since the SPO2 dropped below 65% at 21,000 ft and the subjects were asked to change over to chamber’s MK-17 E regulator for safety. The SPO2 values in Mi-17 IV system (KP-58 regulator) and Ulmer regulator, both coupled with customized TAN mask, were found to be satisfactory and comparable to those of the positive control (Fig 5 (B) and Table 4).
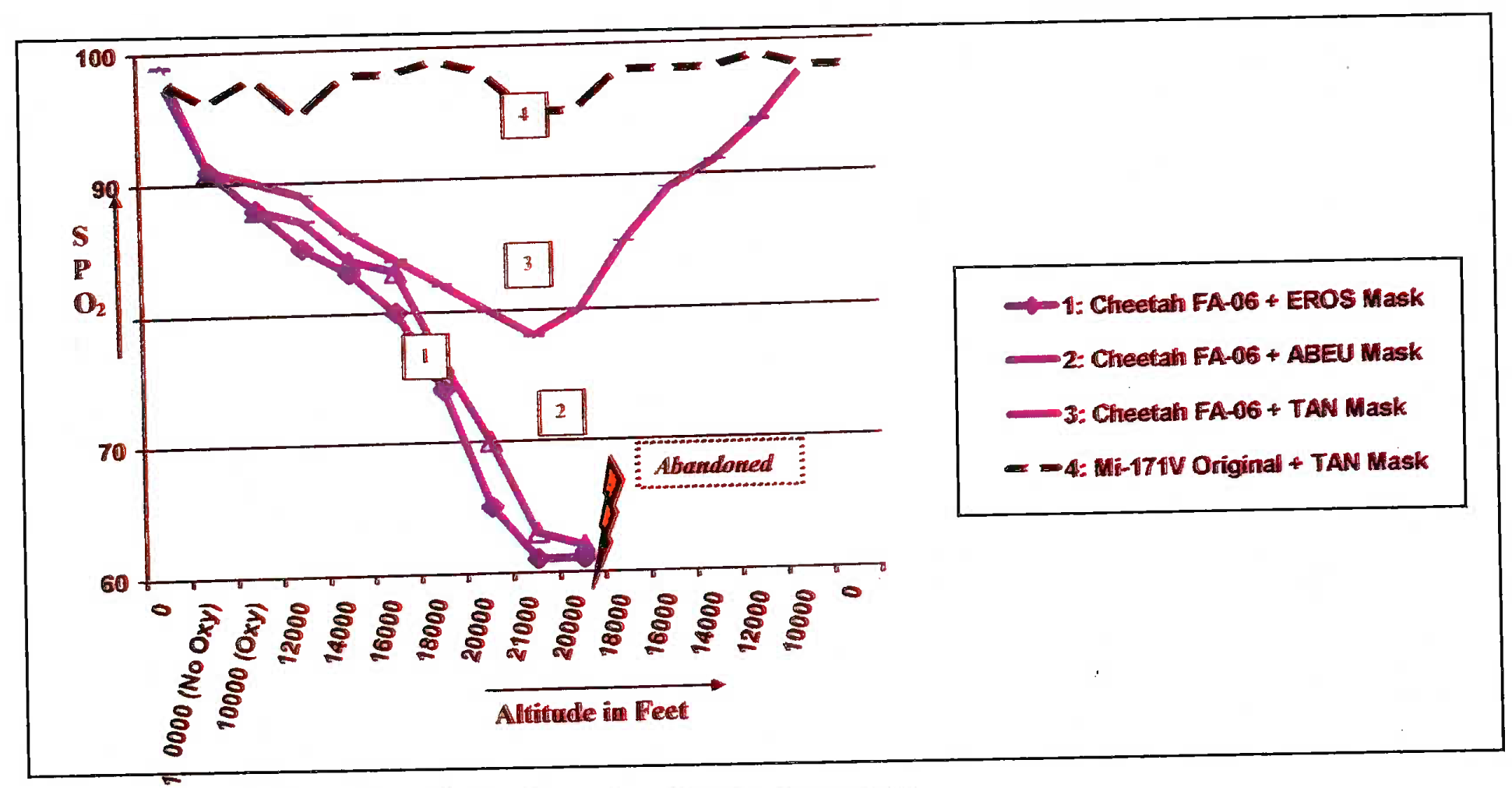
- SPO2 values in% vis-a-vis altitude in feet in different modifications of Cheetah oxygen system (CT 1)
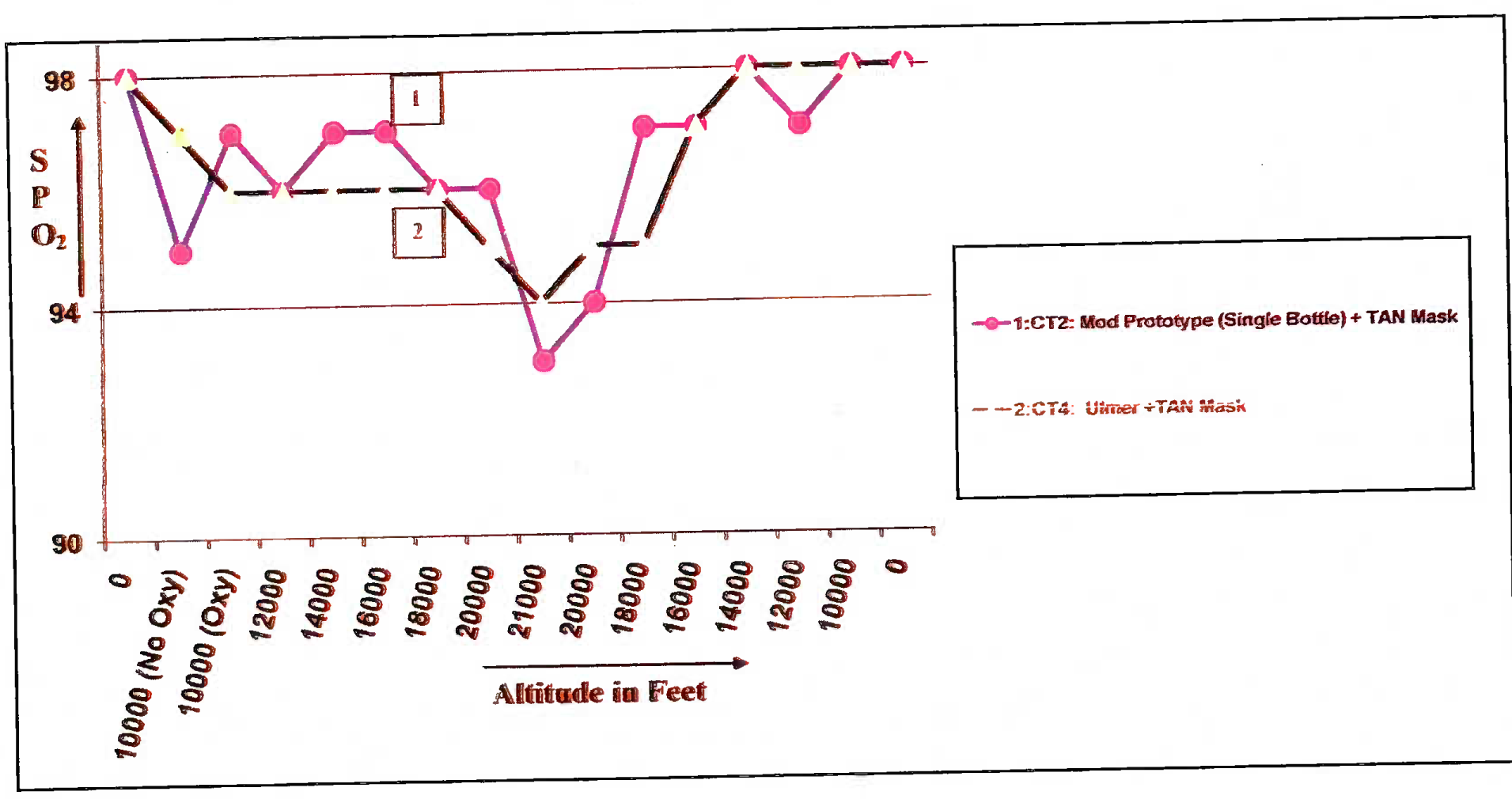
- SPO2 values in% vis-a-vis altitude in feet in different modifications of Cheetah oxygen system (CT 2 & 4)
| Altitude in Ft Modification |
0 | 10000 (No O2) | 10000 (O2) | 12000 | 14000 | 16000 | 18000 | 20000 | 21000 | 20000 | 18000 | 16000 | 14000 | 12000 | 10000 | 0 |
| CT1: Cheetah FA-06 +ERQS Mask | 98 | 91 | 88 | 85 | 83 | 80 | 74 | 65 | - | Recording Terminated and Subjects Asked to Breathe from Mk-l 7E Chamber Regulator | ||||||
| CT 1: Cheetah FA-06 + ABEU Mask | 98 | 91 | 88 | 87 | 84 | 83 | 76 | 70 | 63 | - | ||||||
| CT 1: Cheetah FA-06 +TAN Mask | 99 | 91 | 90 | 89 | 86 | 84 | 82 | 80 | 78 | 80 | 85 | 89 | 91 | 94 | 98 | 98 |
| CT1:M1-171V Qrigmal+TAN Mask | 98 | 96 | 98 | 95 | 98 | 98 | 99 | 98 | 95 | 95 | 98 | 98 | 98 | 99 | 98 | 98 |
| CT 2: Mod Prototype (Single Bottle)+TAN Mask | 98 | 95 | 97 | 96 | 97 | 97 | 96 | 96 | 93 | 94 | 97 | 97 | 98 | 97 | 98 | 98 |
| CT 4: Ulmer+TAN Mask | 98 | 97 | 96 | 96 | 96 | 96 | 96 | 95 | 94 | 95 | 95 | 97 | 98 | 98 | 98 | 98 |
| Mk 17E+ABEU Mask (+vecontrol) | 98 | 95 | 98 | 99 | 98 | 99 | 99 | 98 | 94 | 95 | 96 | 98 | 99 | 99 | 99 | 99 |
(ii) Using the EROS, AB EU and TAN masks with FA-06 regulator, the aircrew persistently had lower than 90% saturation values. The following two factors were primarily responsible for lowered SPO2:
(aa) The high inspiratory resistance on account of 2 inspiratory valves (one in FA-06) and the other in mask itself in ABEU and TAN masks.
(bb) Outboard leakage from ill-fitting masks.
(iii) This drop, with TAN mask, was lesser than that in ABEU mask and was highest with the EROS mask. (e.g. at 20,000 ft during ascent, the SPO2 values for EROS, ABEU and TAN mask with FA-06 regulator were 65%, 70% and 80% respectively). EROS performed the worst due to excessively high mask leakage. TAN mask performed the best due to its highly improved design and anthropometry-based sizing. Deep voluntary breathing by the pilot, on our instructions, recovered SPO2, albeit temporarily. This was not acceptable, as this required a conscious effort on part of the pilot to do intermittent hyperventilation (increased depth of breathing) to counter hypoxia.
(iv) However, it was also realised that the problem of fixed dilution regulator causing reduced oxygen system endurance was not addressed by simply changing the mask. KKO-PC2 system was found to be adequate in maintaining SPO2 when used in conjunction with the customized TAN mask. Based on this trial set, we could directly recommend the use of KKO-PC2 system, in combination with the TAN mask, for use in Mi-17 IV and Mi-17 helicopters.
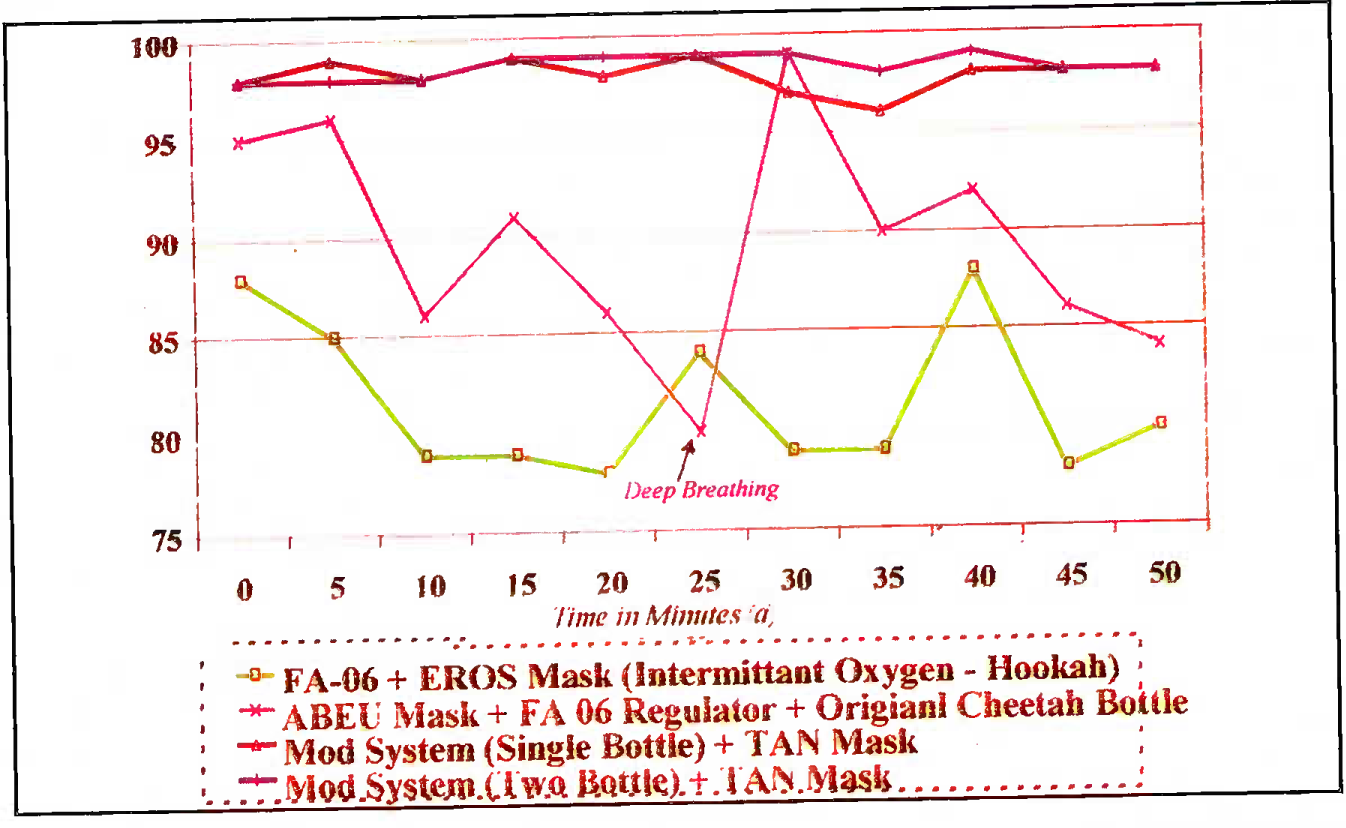
- Inflight SPO2 as a function of time (flight altitude 18,000-21,000 ft)
(c) Chamber Trial 2 (CT 2)
(i) The prototype, comprising of one Cheetah oxygen bottle fitted with KP-75 reducer and KP- 58 regulator, was found to satisfactorily provide SPO2 values above 90% throughout the run (Fig 5 (B), Table 4). This indicated that the modification provided sustained required oxygen concentration at the regulator outlet to sustain normal SPO2 despite the drop in the cylinder pressure. There were no leaks in the modification.
(ii) The mean system endurance in this configuration was found to be 108 minutes, achieved with the, existing Cheetah system (a total of 6 trials). Based on these results, this was the first prototype we considered for the Cheetah.
(d) Flight Trial 2 (FT 2) The prototype, tested in CT 2 (KP-75+KP-58 + customized TAN mask) maintained satisfactory SPO2 in flight (Fig 6). The improved system endurance in flight was also confirmed.
(e) Chamber Trial 3 (CT 3). At present in the Cheetah, each aircrew has 2 interconnected oxygen bottles, one placed behind the instrument panel and one behind the seat. The aircrew manually switches from the empty bottle to the second bottle in-flight when required. The mandate of maximizing the oxygen system endurance necessitated that we maintain a two-bottle configuration for our modification also. This necessitated the development of a suitable junction box connector that would interconnect two bottles fitted with KP-75 reducer and give a single outlet to KP-58 regulator (Fig - 1). This modification was tested in the chamber and found to provide adequate SPO2 The mean endurance of this two bottle prototype was found to be 183 minutes in three trials.
(f) Flight trials 3 and 4 (FT 3 and 4). This was a long phase of trials for ergonomic integration of the 2-bottle modified prototype. In FTC, it was found that the wide KP-75 fitted on the bottle behind the instrument panel obstructed the front view of the aircrew by about 1% of the critical visual field. In FT4, the front bottle was repositioned behind the seat on the outer side but it was found that the aircrew could not satisfactorily read the contents gauge of the bottle fitted near the exit door behind the seat. Further flight trials (FT5) were done at Leh to address the above ergonomic constraints by the engineers. The in-flight Pulse Oximetry studies were done in all these trials to generate further data and the SPO2 values achieved were consistently satisfactory in all the ergonomic variations of the two-bottle prototype (Fig-4).
(g) Chamber Trial 4 (CT 4). Ulmer demand diluter automatic regulator (model 7893A), along with the existing Oxygen bottle, reducer and TAN mask was tested. It is pertinent to bring out that the same regulator attached to Ulmer mask was tested at IAM, IAF in 1998 for the Army Aviation. During this testing, the Oxygen enrichment at the inspiratory valve was tested using Servomax™ equipment. In this study however, no Pulse Oximetry studies were done [3]. Moreover, there was a need to evaluate the compatibility of this regulator with the TAN mask. In CT 4, we were able to generate that data. This configuration was shown to maintain the SPO2 values above 90% till an altitude of 20,000 ft (total of 2 trials) (Fig - 5 (B), Table - 4).
(h) Flight trial 5 (FT 5). The aim of this final trial was to compare the prototype developed in CT3 with that developed in CT4. The SPCf values in mod prototypes tested in CT3 and CT4 were found to be > 90% during flight-testing. In these comparative trials, Ulmer regulator was found to be ergonomically more convenient than the double-bottle configuration.
Based on physiologic and ergonomic considerations in the above trials, we were able to recommend the components of the Oxygen systems for Mi-17 and Cheetah helicopters. These components are summarized at Table - 5.
| Cheetah F | di-17/Mi-17 IV | |
|---|---|---|
| Bottle Pressure | 150 bar | 210 kg/cm2 |
| Reducer | EROS | KP-75 |
| Regulator | Ulmer | KP-58 |
| Mask | TAN | TAN |
| Helmet | Integrated | Leather inner |
| Microphone | Mask integrated | Mask integrated |
Oxygen System Evaluation - Engineering vis-a-vis Aeromedieal issues Traditionally, the oxygen system testing methods is done in accordance with the relevant laid down standards (e.g. testing methods by OEM, MEL-STD, Defstan etc). This requires a well-set testing lab that can measure all the recommended parameters on ground. Interestingly, the Mil Standards do not speedy any testing of the oxygen system at altitude, simulated or actual, where the oxygen dilution and delivery actually starts. In the field, such sophisticated test benches are not available. Moreover, a field investigator would be more interested in preliminarily assessing the user’s complaints related to his system, find the hypoxia causing potential of a defective oxygen system and offer solutions. In this regard, the use of a portable system, sensitive enough to measure hypoxia-related parameters (viz SPO2 and pulse rate), is considered important.
Pulse Oximetry [4, 5, 6]
Nomin® (model 8600) Oximeter probe shines red (660 nanometers) and infrared (910 nanometers) light through the tissue and detects the fluctuating signals caused by arterial blood pressure pulses. The instrument determines the Oxygen saturation from the difference in absorption of these colour bands during a pulse and in its absence. Since other factors such as steady venous blood flow, colour of skin, etc., do not cause fluctuations in the colour, they do not affect the saturation readings. Mathematically, SPO2 is calculated as follows:
US Air Force has accepted Nonin® 8600 model with its internal battery for in-flight usage. The Oximeter is calibrated during manufacturing and automatically checks its internal circuits when turned on [4]. Since repeated calibration is not required, the equipment is a highly convenient gadget for field use and by corollary for aviation research.
The SPO2 values below 70% are unreliable in Pulse Oximeters due to the logarithmic scale of calculation. Hence, in the chamber / in flight trials, 65% SPO2 was used as cut off in data recording. The subject was asked to use control Oxygen delivery system (MK 17 E regulator in chamber & KKO-PC2 onboard system carried in flight).
In our study, ambient cold temperature at 18000 ft (-15 °C at Leh in winter months) was the only possible factor that could vitiate the SPO? readings. We however faced no problems in this regard as the aircrew were wearing winter flying gloves that maintained the skin temperature over the fingers, aircraft cockpit heater increased the temperature above ambient and the aircrew were asked to briefly rub hands prior to recording.
Heart rate increases on exposure to hypoxia and has been used in Oxygen system assessment studies. In our trials, we did not find heart rate changes to be reliable and repeatable indicator. Our recommendations were hence primarily based on SPO2 data.
Thus, Pulse Oximetry is an important tool available to the field researcher that can provide relevant inputs for defect / fitness analysis in subsequent bench testing. With this background philosophy, we decided to use Pulse Oximetry in our trials.
Conclusions
Pulse Oximetry served as a guideline in determining the physiologic adequacy of the prototype combinations in the developmental process. This well-known technique was found to be highly dependable in our trials. At the end of the trials, we were able to provide satisfactory solutions, well tested in flight and approved by the operating pilots.
References
- Problems and Solutions of Oxygen Systems of Helicopters of IAF Flying at High altitude.
- Military Specification MIL-R-83178A (USAF): Aircraft Regulators.
- The elusive oxygen mask-helmet system for Cheetah helicopter: A cause for erosion of accepted norms in the field. Ind J Aerospace Med. 1999;43(2):24-7.
- [Google Scholar]
- Theory of Operation. :42-3. Nonin® Instruction and Service Manual, Models 8600 & 8600M Pulse Oximeters
- Recommended use of pulse Oximeter in Aviation. Sited at web page http://www.avweb.com/news/aeromed/181896-l.html
- ‘Pulse Oximetry’ Practical Procedures. 1995;5(Article 2):1. Available at www.nda.ox.ac.un/wfsa/htm/u05/u05/003.htm






