Translate this page into:
Hypoxia signature: A useful tool for hypoxia recognition among aircrew

*Corresponding author: Dr S Kasture, MBBS, MD (Aerospace Medicine), Tambaram, Chennai - 600045, Tamil Nadu, India. shivani.kasture30@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Kasture S, Nataraja MS, Mohapatra SS, Sinha B. Hypoxia signature: A useful tool for hypoxia recognition among aircrew. Indian J Aerosp Med 2021;65:81-85.
Abstract
Introduction:
Hypoxia, often referred to as “silent killer,” a common aeromedical stressor in aviation, may have catastrophic events in-flight unless recognized well in time. On exposure to hypoxia, an individual manifests a specific spectrum of symptoms referred to as “hypoxia signature.” The present study was conducted to assess the manifestation of “hypoxia signature” on repeated exposure to simulated hypobaric hypoxia for its potential usage as a tool for hypoxia recognition.
Material and Methods:
Twenty-two healthy adult volunteers were subjected to a simulated altitude of 22,000 feet for a duration of 5 min in the hypobaric altitude chamber. The symptoms experienced by the participants at the said altitude were recorded using a questionnaire. The heart rate (HR) and oxygen saturation (SpO2) were recorded during the exposure. The hypoxia exposure was repeated two more times with a minimum interval of 3 weeks between each. Paired t-test was used to compare the mean values of physiological parameters (HR and SpO2) between ground level and 22000 feet recorded in all the three exposures. The hypoxia symptoms and their severity reported during the exposures were compared with those of recalled symptoms (reported after 3 weeks of exposure) using McNemar test and Wilcoxon Signed Rank test, respectively.
Results:
Paired t-test revealed a statistically significant increase in HR and fall in SpO2 with rise in altitude from ground level to 22000 feet. The three most common symptoms consistently observed were lightheadedness, thinking slow, and warm feeling. The common hypoxic symptoms and their severity scores reported at 22,000 feet compared with recalled counterpart during subsequent exposures did not reveal any significant differences (P > 0.05).
Conclusion:
There was a high degree of similarity in the frequency and severity score of symptoms between acute exposure to hypobaric hypoxia and recall indicating evidence of repeatability of symptoms across the three sessions of hypoxia exposure within the individuals. This brings out the usefulness of “hypoxia signature” as a tool for hypoxia recognition and its application in hypoxia indoctrination and training for aircrew.
Keywords
Hypobaric hypoxia
Hypoxia signature
Repeatability
Hypoxia indoctrination and training
INTRODUCTION
Acute hypobaric hypoxia has been the single most serious hazard of flight during high altitude flying operation, and it remains a serious threat even today.[1] Loss of aircraft cabin pressurization and failure of oxygen system accounts for the majority of causes of in-flight hypoxia.[1,2] While the effects of sudden or rapid loss of cabin pressurization are obviously apparent to the aircrew, slow or unrecognized depressurization and failure of oxygen systems are subtle in nature, and are often not easily recognized.[3] This has earned hypoxia its dangerous reputation as the “silent killer.” The order of appearance of signs and symptoms that accompany hypoxia and their severity are related to the exposure period and the rate and degree to which the blood oxygen tension is lowered.[1]
Further, depending on the level of altitude, a relatively fixed period of time is available between reduction in partial pressure of oxygen and development of specified degree of decline in mental performance, commonly referred to as “Time of Useful Consciousness.”[1] This is the window period in which the aircrew has to recognize the hypoxia symptoms and take corrective actions.
There is evidence to suggest that, occurrence of hypoxic symptoms, the order and speed of their appearance, and their severity for an individual usually remains consistent over time.[4] This is commonly referred to as “hypoxia signature” and is unique to the individual, just like one’s signature. It is thus essential for an aircrew to identify his hypoxia signature as a part of training and indoctrination so that early corrective actions can be instituted in case hypoxia occurs in-flight. The present study was conducted to assess the evidence of hypoxia signature on acute exposure to hypobaric hypoxia. The objective was to identify the individual hypoxia symptoms and demonstrate its reproducibility so as to recommend whether hypoxia signature can be used as a useful tool for hypoxia training and aeromedical indoctrination. There are other studies who have reported hypoxia signature;[4-7] however, this study was undertaken to examine hypoxia signature in Indian scenario.
MATERIAL AND METHODS
Subjects
A total of 22 non-aircrew healthy adult volunteers (Age = 32.63 ± 4.34 years, height = 169.86 ± 8.81 cm, and weight = 70.90 ± 10.53 kg) participated in the study. Informed consent was obtained from the participants after explaining the procedure in detail. The study was approved by the Institute Ethics Committee.
Materials
The Hypobaric Altitude Chamber in the Department of High Altitude Physiology and Hyperbaric Medicine was used to simulate hypobaric hypoxia. Heart rate (HR) and oxygen saturation (SpO2) were two physiological parameters recorded during the experimentation. SpO2 was measured using pulse oximeter. A questionnaire consisting of 24 hypoxia symptoms (e.g., light-headedness, Thinking slow, Warm feeling, Tingling, and Dizzy) was given to all participants and were asked to grade the severity of the symptoms experienced by them during exposure to hypoxia on a scale of 1–5.[8]
Procedure
All participants breathed 100% oxygen for 30 min before exposure to hypoxia. An ear clearance run was conducted by ascent from ground level till 10,000 feet and then descent to ground level @ 3000 feet/min to exclude any participant’s risk of barotrauma. Participants were then exposed to a simulated altitude of 22,000 ft for 05 min. The ascent and descent rates were kept at 3000 ft/min. The profile of hypoxia exposure is depicted in Figure 1. During the run, participants were on mask. At desired altitude of 22,000 feet, the participants were instructed to remove their masks and breathe cabin air. They were instructed to note down the occurrence of symptoms with their severity in the questionnaire. HR and SpO2 were continuously monitored. If the SpO2 of any participant fell below 65%, the exposure of hypoxia was terminated by asking the respective participant to resume breathing 100% oxygen through the mask immediately. The same protocol was repeated on two more occasions for a total of 3 times with an interval of 3 weeks between the two consecutive exposures. During the repeat exposures (after 3 weeks of acute exposure), the participants were asked to recall the symptoms and grade with severity experienced by them during their previous exposure, that is, “recalled symptoms.”
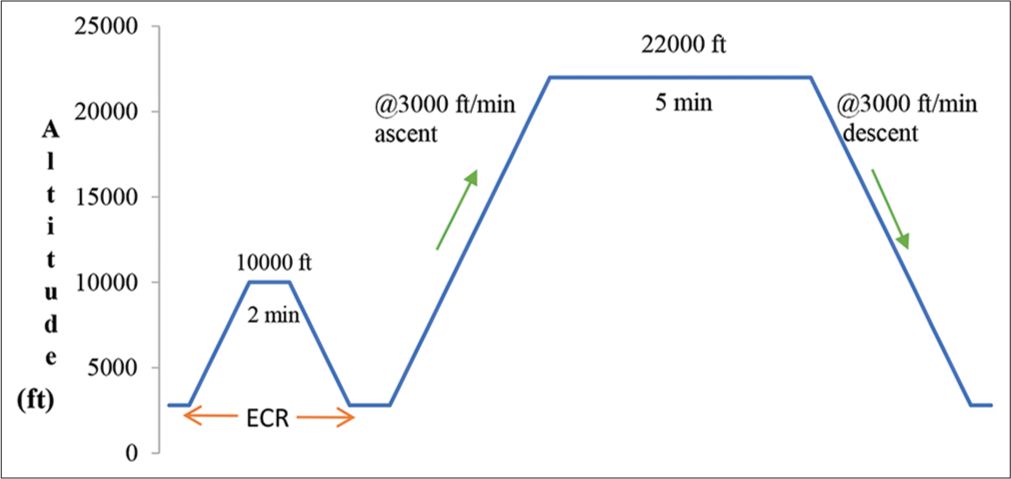
- Profile of simulated acute hypobaric hypoxia exposure.
Statistical analysis
The collected data were subjected to descriptive and analytical statistical tests. The frequencies of three commonly occurring symptoms at 22,000 feet reported by the participants on all 3 days of exposure were compared with counterpart symptoms obtained from recall memory of participants using McNemar test. The severity scores of same common symptoms observed at 22,000 feet were compared using Wilcoxon signed rank test. Paired t-test was applied to determine the effect of altitude (independent variable) on HR and SpO2 (dependent variable). Level of significance was set at P < 0.05. The statistical tests were done using Statistica 9.0®.
RESULTS
A total of 19 (86.6%), 17 (77.2%), and 18 (81.8%) of the participants reported hypoxia symptoms on 1st, 2nd, and 3rd exposure to 22,000 feet, respectively. The three common symptoms reported at 22,000 ft were light headedness, thinking slow, and warm feeling. The occurrence of these three common symptoms on acute exposure and number of participants who could recall these symptoms are presented in Table 1. McNemar’s test did not reveal statistically significant difference (P > 0.05) between the frequency of the three common symptoms reported during the acute exposure and the recalled symptoms on all three exposures. Wilcoxon signed-rank test did not reveal any statistically significant difference between the severity of symptoms experienced between acute exposure and that of recalled, on all the three hypoxic exposures at 22,000 feet [Table 2].
| Three common symptoms | 1stExposure | 2nd Exposure | 3rd Exposure | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Acute | Recalled | P-value | Acute | Recalled | P value | Acute | Recalled | P value | |
| Light headedness | 9 | 8 | 1.00 | 10 | 6 | 1.00 | 10 | 6 | 1.00 |
| Thinking slow | 8 | 4 | 0.28 | 6 | 7 | 0.12 | 10 | 11 | 0.21 |
| Warm feeling | 7 | 7 | 1.00 | 7 | 7 | 1.00 | 5 | 6 | 1.00 |
| Three common symptoms | 1st Exposure | 2nd Exposure | 3rd Exposure | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Acute | Recalled | P value | Acute | Recalled | P value | Acute | Recalled | P value | |
| Light headedness | 4 | 5 | 0.76 | 4 | 3 | 0.73 | 2 | 4 | 0.71 |
| Thinking slow | 4.50 | 5.14 | 0.08 | 6 | 3 | 0.33 | 8 | 4.63 | 0.07 |
| Warm feeling | 1.50 | 2.25 | 0.41 | 2.50 | 3.13 | 0.15 | 2 | 2 | 0.56 |
Paired t-test revealed that the mean HR values at 22,000 feet were significantly higher than ground level on all three exposures [Table 3]. SpO2 of 07 individuals on 1st exposure, 06 individuals on 2nd exposure, and 06 individuals on 3rd exposure fell below 65% during the course of hypoxia exposure. Paired t-test revealed a significant fall in the mean SpO2 values at 22,000 feet compared to ground level on all the three exposures [Table 4].
| HR (bpm) | Altitude | n | Mean | SD | t | df | p value |
|---|---|---|---|---|---|---|---|
| Day 1 | Ground | 15 | 79.26 | 8.96 | -6.06 | 14 | < 0.001 |
| 22K ft | 101.60 | 14.38 | |||||
| Day 2 | Ground | 16 | 81.81 | 5.12 | -7.66 | 15 | < 0.001 |
| 22K ft | 99.12 | 10.67 | |||||
| Day 3 | Ground | 16 | 73.62 | 7.79 | -7.85 | 15 | < 0.001 |
| 22K ft | 96.37 | 12.87 |
HR: Heart rate
| SpO2(%) | Altitude | n | Mean | SD | t | df | P value |
|---|---|---|---|---|---|---|---|
| Day 1 | Ground | 15 | 98.80 | 0.56 | 17.36 | 14 | <0.001 |
| 22K ft | 73.66 | 5.44 | |||||
| Day 2 | Ground | 16 | 98.75 | 0.57 | 13.16 | 15 | <0.001 |
| 22K ft | 72.43 | 7.70 | |||||
| Day 3 | Ground | 16 | 98.87 | 0.34 | 15.46 | 15 | <0.001 |
| 22K ft | 71.87 | 6.93 |
SpO2: Oxygen saturation
DISCUSSION
Sudden exposure to altitude beyond 10,000 feet exposes an aircrew to effects of hypoxia. Consequences of exposure to hypoxia range from trivial physiological symptoms to total incapacitation depending on the altitude. Exposure to more than 20,000 feet altitude is known to result in critical stage of hypoxia, which, unless promptly identified and corrected, may result in catastrophic event.[1] In-flight hypoxia, often referred as a silent killer, needs to be recognized early and corrective actions need to be implemented. The best part is that manifestations of hypoxic symptoms, and their severity usually remains consistent for an individual typically referred to as “hypoxia signature.”[4] Examination of recall and reproducibility of this hypoxia signature on repeated simulated acute exposure to an altitude of 22,000 feet was the desired objective of the present study.
As per the recommended practice in the IAF,[9] the hypoxia indoctrination is undertaken at 25,000 feet. However, we chose an altitude of 22,000 feet in the present study due to several factors; (a) the incidence and severity of hypoxia become evident at a cabin altitude of 22,000 feet and above.[10] (b) The TUC at 22,000 feet is approximately 10 min thus allowing sufficient time for experiencing and recognizing hypoxia symptoms without manifesting significant impairment in pilot performance.[11] (c) The risk of DCS increases significantly beyond this altitude.[12] (d) The cabin altitude in low differential pressure cabin such as combat fighter aircraft is maximum allowed up to 22,000 feet.[10] Pre-breathing with 100% oxygen for a period of 30 min was given to all participants to eliminate the risk of DCS as per the recommended guidelines.[13]
The physiological parameters, namely, HR and SpO2 on acute exposure to 22,000 feet, on all the three occasions, showed changes as physiologically expected. The HR showed a significant increase [Table 3] whereas, the SpO2 was found to be significantly decreased [Table 4]. These changes can be attributed to activation of peripheral chemoreceptors mechanisms as a result of decrease in oxygen tension in breathing air consequent to exposure to hypoxia.[14] Similar findings have also been reported by other studies.[8,15,16]
In the present study, the number of symptoms reported by participants on all the three occasions was comparable. However, out of the 22 participants, three individuals did not experience any symptoms on any of the occasions. These findings suggest the possibility of individual variations in hypoxia tolerance and the occurrence of hypoxia symptoms. Other studies have also reported similar findings in which few individuals did not experience any symptoms on exposure to hypoxia.[4,5,17]
The commonly reported symptoms on exposure to 22,000 ft noticed were “light-headedness,” “thinking slow” and “feeling warm.” These findings are similar to that of Woodrow et al. who also reported the most common symptom as light-headedness in both recalled and refresher training sessions.[6] In a study by Tu et al., the reported common symptoms were poor concentration (30.5%), impaired cognitive function (20.5%), visual disturbances (16.4%), hot flushes (15.85%), and paraesthesia (12.6%).[7] Two of the commonly reported symptoms in our study were similar to those reported by Tu et al. These were thinking slow (subset of impairment in cognitive function) and warm feeling (a synonym of hot flushes). The difference in other reported symptoms in both the studies can be attributed to “different altitude of exposure” and “number of subjects,” that is, 25000 ft and 341, respectively, in Tu et al. study whereas, it was 22000 ft and 22 in our study.
A comparison between the three commonly reported symptoms (light-headedness, thinking slow, and warm feeling) experienced at 22,000 feet on all the three occasions with recalled symptoms revealed similarities between the two. Another important observation in our study was absence of significant difference in the severity score of symptoms between acute exposure and recall symptoms. Studies by Smith and Johnston et al. have also reported a high level of agreement between symptoms in recalled and fresh training session.[4,5] However, few authors[6,7] have reported significant differences in hypoxia symptoms between the two sessions. The possible reason given by Woodrow was that either no subject could exactly remember the symptoms experienced in the previous training or control measure could have been different affecting results during hypoxia events.[6] Tu et al. also felt that memory of hypoxia symptoms might vary among the individuals and fade over a period of time.[7] Notwithstanding the above, the findings of our study point towards potential benefit of recognizing individual specific hypoxic symptoms for aeromedical indoctrination of aircrew and hypoxia training.
The exact mechanism of memory storage in the brain is still not completely understood. As per long-term potentiation model, certain synapses undergo long-lasting increase in efficacy when they are repeatedly used leading to alteration of gene expression and synthesis of new proteins. These new proteins are responsible for increase in number of synapses that have been demonstrated after long-term memory formation.[18] The hypobaric hypoxia simulated in the present study and repeated for three occasions with a minimum gap of 3 weeks between them, possibly lead to long-term memory formation, enabling participants to recall hypoxia symptoms faultlessly. Thus, it can be inferred that refresher hypoxia training would benefit the aircrew in recognizing symptoms of hypoxia. Thus, the phenomenon of “hypoxia signature” reported by Smith[4] in his study, could also be established in our study by repeated simulated exposures to acute hypoxia of 22,000 feet. This has direct implications in hypoxia indoctrination and training of aircrew.
CONCLUSION
The findings of present study revealed a high degree of similarity in the frequency and severity scores of hypoxia symptoms reported during the exposure to simulated altitude of 22,000 feet and the symptoms recalled following subsequent exposures to same altitude. Thus, it can be inferred that the symptoms experienced by participants on acute exposure to hypoxia remain stable over a period of time. In addition, 22,000 feet altitude can be used as the preferred altitude for demonstration of hypoxia signature considering adequate time of useful consciousness, less risk of DCS, and the realistic maximum cabin altitude maintained in military fighter aircraft.
Declaration of patient consent
The authors certify that they have obtained all appropriate consent from the participants.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Hypoxia and hyperventilation In: Gradwell DP, Rainford DJ, eds. Ernsting's Aviation and Space Medicine (5th ed). Boca Raton: CRC Press; 2016. p. :49-64.
- [CrossRef] [Google Scholar]
- In-flight hypoxia incidents in military aircraft: Causes and implications for training. Aviat Space Environ Med. 2003;74:169-72.
- [Google Scholar]
- Examining Hypoxia: A Survey of Pilots' Experiences and Perspectives on Altitude Training Oklahoma City OK: Federal Aviation Administration. In: Civil Aeromedical Institute. 2003.
- [CrossRef] [PubMed] [Google Scholar]
- Hypoxia symptoms in military aircrew: Long-term recall vs. acute experience in training. Aviat Space Environ Med. 2008;79:54-7.
- [CrossRef] [PubMed] [Google Scholar]
- Hypoxia training: Symptom replication in experienced military aircrew. Aviat Space Environ Med. 2012;83:962-7.
- [CrossRef] [PubMed] [Google Scholar]
- Recollection of hypoxia symptoms between training events. Aviat Space Environ Med. 2011;82:1143-7.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of hypobaric hypoxia symptoms between a recalled exposure and a current exposure. PLoS One. 2020;15:e0239194.
- [CrossRef] [PubMed] [Google Scholar]
- Hypobaric and normobaric hypoxia training in aircrew: A comparative study. Indian J Aerosp Med. 2013;57:28-36.
- [Google Scholar]
- A retrospective analysis of altitude chamber training protocols and the adverse effects at Aero Medical Training Centers in the Indian Air Force: 1998-2003 In: Paper Presented at 52nd International Congress on Aviation and Space Medicine 2004. 2004.
- [Google Scholar]
- Oxygen systems, pressure cabin and clothing In: Gradwell DP, Rainford DJ, eds. Ernsting's Aviation and Space Medicine (5th ed). Boca Raton: CRC Press; 2016. p. :79-129.
- [CrossRef] [Google Scholar]
- Respiratory physiology and protection against hypoxia In: Davis JR, Johnson R, Stepanek J, Forgarty JA, eds. Fundamentals of Aerospace Medicine (4th ed). Philadelphia, PA: Lippincott Williams and Wilkins; 2008. p. :20-45.
- [Google Scholar]
- Physiology of decompressive stress In: Davis JR, Johnson R, Stepanek J, Forgarty JA, eds. Fundamentals of Aerospace Medicine (4th ed). Philadelphia, PA: Lippincott Williams and Wilkins; 2008. p. :46-82.
- [Google Scholar]
- Altitude-induced Decompression Sickness: Tiny Bubbles, Big Troubles AM-400-95/2 United States: FAA Civil Aeromedical Institute, Aeromedical Education Division; 1995. p. :1-6.
- [Google Scholar]
- Exercise in hypoxic environments: The mechanism remains elusive. J Physiol. 2003;550:335.
- [CrossRef] [PubMed] [Google Scholar]
- A Comparative Study of Measurement of SpO2 between Transmittance vis-à-vis Reflective Type of Pulse Oximeter in Simulated Hypobaric Hypoxia and Immediately after Simulated Hyperbaric Hyperoxic Exposure Bangalore: Rajiv Gandhi University of Health Sciences; 2020.
- [Google Scholar]
- Effects of acute hypoxia on heart rate variability, sample entropy and cardiorespiratory phase synchronization. Biomed Eng Online. 2014;13:73.
- [CrossRef] [PubMed] [Google Scholar]
- Aviation Medicine for Aircrew (2nd ed). Adelaide, Australia: Institute of Aviation Medicine; 2002. p. :41-8.
- [Google Scholar]
- Consciousness, the brain and behaviour In: Vander's Human Physiology: The Mechanisms of Body Function (12th ed. Ch. 8). New York: McGraw-Hill; 2008.
- [Google Scholar]






