Translate this page into:
Cross tolerance of two aviation stresses-Acute heat stress and hypoxia
-
Received: ,
Accepted: ,
Abstract
Simultaneous exposure to thermal and hypoxic stress is common in military flying. 10 healthy non–smokers in the age group of 25-32 were evaluated at ground level and at 18,000 ft to see the physiological effects of induction of Heat Shock Protein-70 (HSP-70) on performance in a working memory task in hypoxia in a within subject experimental design. Subjects were evaluated for computer based visual memory test consisting of correct responses, incorrect responses and reaction time in Explosive Decompression Chamber (EDC) both at ground level for baseline readings and at a simulated altitude of 18,000 ft after 30 minutes of hypobaric-hypoxic exposure. The main effect of HSP and interaction effect of HSP and altitude were observed on correct responses and reaction time in the present study. The number of correct responses at ground level and at 18,000 ft was 29.48±1.038 and 28.57±1.159 respectively in absence of HSP induction and the responses were 30.34±0.920 and 30.90±0.991 in presence of HSP induction. Reaction time was 0.825±0.073 sec, 0.857±0.660 sec in absence of HSP and 0.799±0.572 sec, 0.774±0.560 sec in presence of HSP induction at ground level and at 18,000 ft respectively. The present study indicates that the HSP induction has improved the performance in a working memory task in hypoxia.
Keywords
Hypoxia
Heat stress
Heat shock protein
Reaction time
Correct responses
Introduction
Thermal stress and hypoxic stress are very common in military flying. The effect of hypoxia in causing impairment of human performance and cognitive abilities is also well known [1,2]. Psychomotor deficits are a prominent feature in subjects exposed to hypoxia [3].
It is reported that effect of pre-induction to one stress can significantly modify the physiological responses during subsequent exposure to a second stress. Study indicates that the survival of neurons under ischemic condition seemed to be related to the amount of Heat Shock Protein-70 (HSP-70) induced [4]. In Mice model, it was observed that over expression of HSP-70 in the brain protect them against cerebral infarction following cessation of blood supply [5]. HSP-70 protects murine astrocytes from glucose deprivation injury [6]. Gene therapy with HSP-72 is neuroprotective in rat models of stroke and epilepsy [7]. The expression of HSP-70 is an important reaction in the brain, and it has close relationship with brain ischemic injury. HSP-70 induction during +Gz stress can protect the nerve cells after brain ischemia [8].
One of the first physiological functions associated with the stress-induced accumulation of the inducible HSP-70 was acquired thermo-tolerance, which is defined as the ability of a cell or organism to become resistant to heat stress after a prior sub-lethal heat exposure [9].
In the light of the above, it was hypothesized that the pre-induction of HSP-70 in acute heat exposure may confer protection in subsequent hypoxia exposure. No study is available on the effect of induction of HSP-70 on performance in a working memory task in hypobaric hypoxia. The present study was conducted to examine the effect of pre-exposure to heat stress on working memory and reaction time response task at an altitude of 18,000 ft in 10 normal healthy non-smoker subjects.
Material and Methods
10 clinically healthy male non-smoker voluenteer subjects in the age group of 25-32 years were selected for this study. All the subjects were explained about the study protocol in a language they understood and they were also apprised of the possible harmful effects of the study they were undergoing. Informed consent was taken from all the subjects before exposing them to heat and hypoxic stress. They were divided into 2 groups, A & B having 5 subjects in each group. Age, height and body weight of the subjects were 30.0±0.67 yrs, 175± 10.03 cms and 73±13.42 kg respectively. Each subject visited the laboratory twice. The design of the study was within subject experimental design.
Group A - On the first visit, the subject was exposed to acute heat stress of dry bulb temperature (Tdb) of 55oC and wet bulb temperature (Twb) of 30oC for 60 minutes. The heat exposure was carried out from 0800 h to 0900 h in a thermal chamber. Subjects were then taken to the Explosive Decompression Chamber (EDC) 6 hrs after heat exposure. They were evaluated with a computer based visual memory task described below. The chamber altitude was gradually raised to an altitude of 18,000 ft within 15 minutes by decompression. Subjects were re-evaluated with the computer based task after 30 minutes of exposure to hypobaric hypoxia. The subjects did not breathe any supplemental oxygen throughout the hypoxic exposure. The chamber altitude was reduced slowly by recompression (descent rate of 500 ft/min) after the task was completed. Subjects visited the laboratory for the 2nd time after one week of the first visit. Working memory task and reaction time response of the subjects were evaluated at ground level and at 18,000 ft in the EDC chamber without any induction of HSP-70.
Group B - The sequence of exposure to heat and hypoxia in this Group was hypoxia on the first day and on another day heat exposure first followed by exposure to hypoxia 6 hrs later on the same day. There was a gap of 7 days between hypoxia exposure and heat-hypoxia exposure.
Performance of working memory task
The procedure involved administration of PC based working memory task. In the working memory task, subject was to identify a target stimulus in an array of similar stimuli. The stimulus was one of ninety geometrical figures of different shapes and of the size of approximately 3.8 x 2.5 cm. The arrays comprised of four such stimuli (two on the left and other two on the right of the array) arranged horizontally with an inter-stimulus separation of 0.6 cm. One of these stimuli might be (or might not be) be the target stimulus. The subject was asked to respond by different key presses, if the target appeared in the right or the left of the array or it did not appear in the array at all. These entire occurrences were kept equiprobable in the task programme. Performance in working memory task i.e. correct responses, incorrect responses and reaction time before and after induction of HSP at ground level and at simulated 18,000 ft was then evaluated by a computer based visual memory test. Characteristics of the task are as shown in Table 1.
| Type of delays | Time |
|---|---|
| Pre-target delay | 500 msec |
| Exposure time of target | 200 msec |
| Pre-array delay | 500 msec |
| Exposure time of pre-array | 400 msec |
| Inter stimulus delay | 500 msec |
Number of correct, incorrect responses, total number of stimuli presented and response time for the instances wherein the target appeared in the right or left of the array or when the target did not appear in the array were computed automatically by the programme and displayed at the end of the task. Correct responses and incorrect responses on the right side, left side, absent correct and absent incorrect responses and reaction time in sec were noted down after the task was completed. The task was administered for 5 minutes. Performance data is normalised for 100 responses from the total number of responses made by the subject in four different experimental conditions during 5 minutes task. Subjects were not allowed to practice the tasks. They were only familiarized with the task procedure.
Statistical Analysis
The data was first checked for normality by Shapiro Wilk’s ‘W’ statistic. Data followed a normal distribution pattern. 2 factors repeated measure ANOVA was used to analyse the data. The two factors were HSP with two levels i.e. before induction and after induction and level of altitude i.e. ground level and 18,000 ft. Post hoc analysis of the data was carried out by using LSD test after the significant outcome from Repeated measure ANOVA.
Results
Performances in the working memory task of the individual were assessed by evaluating correct responses, incorrect responses, and reaction time at ground level and 18,000 ft before and after induction of HSP. Performance data for reaction time responses on the right side, left side and non-appearance was pooled together and analysed. Number of correct responses on the right side, left side and non-appearance was also pooled together and analysed. Number of Correct responses and reaction time before and after induction of HSP at ground level and at 18,000 ft are as shown in Table 2.
| Without induction of HSP | With induction of HSP | |||
|---|---|---|---|---|
| Ground [A] | 18,000 ft [B] | Ground [C] | 18,000 ft [D] | |
| Correct responses (in numbers) | 29.48±1.038 | 28.57±1.159 | 30.34±0.920 | 30.90±0.991 |
| Reaction time (in sec) | 0.825±0.073 | 0.857±0.660 | 0.799±0.572 | 0.774±0.560 |
Correct Responses were standardized for 100 responses to account for variation in the number of attempts made by the individuals in computer based visual memory task administered for 5 minutes. Analysis of the Parameter incorrect responses was not carried out as this parameter is reciprocal to correct responses. Result of statistical significance of main effect of HSP, main effect of altitude and interaction effect of HSP x with altitude on correct response and reaction time are shown in Table 3. Post hoc analysis of the data was carried out by LSD test and as shown in Table 3.
| Main Effect of HSP | Main Effect of Altitude | Interaction Effect of HSP x Altitude | |
|---|---|---|---|
| Correct Responses | F=29.862, p=0.000398 | F=0.306, p=0.593 | F=9.656, p=0.0126 |
| Reaction Time | F=32.710; p=0.000287 | F=0.361;p=0.563 | F=44.237; p=0.000094 |
2-factor repeated measure ANOVA was done. Bold numbers represent significant effect. Post hoc analysis was carried out for individual comparison by LSD test
Changes in working memory task
The correct responses at ground level and at 18,000 ft before and after HSP-70 induction are shown graphically in Fig. 1. The reaction time for responses is shown in Fig. 2.
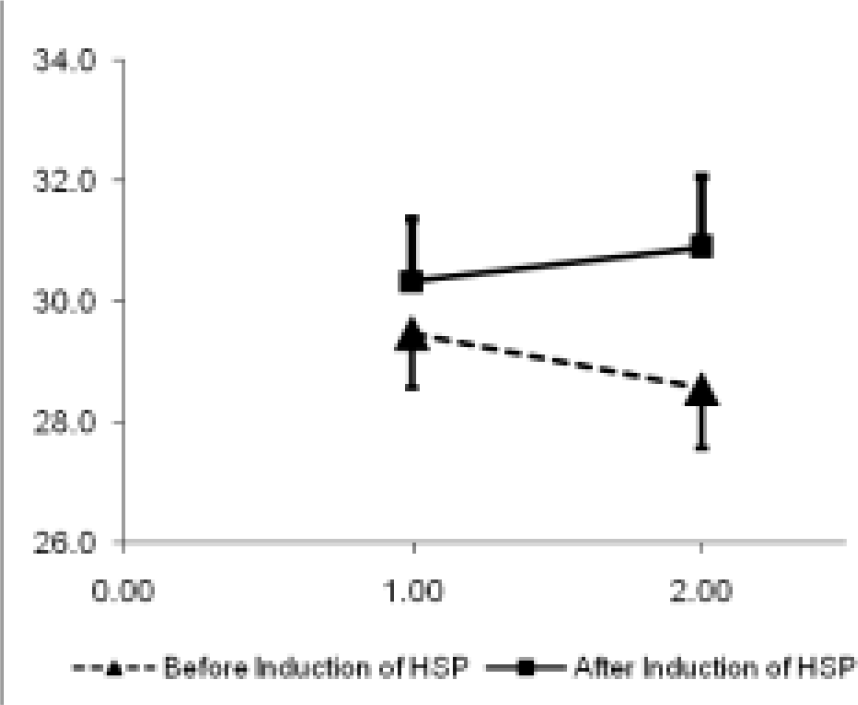
- Correct responses at ground and at 18,000 ft
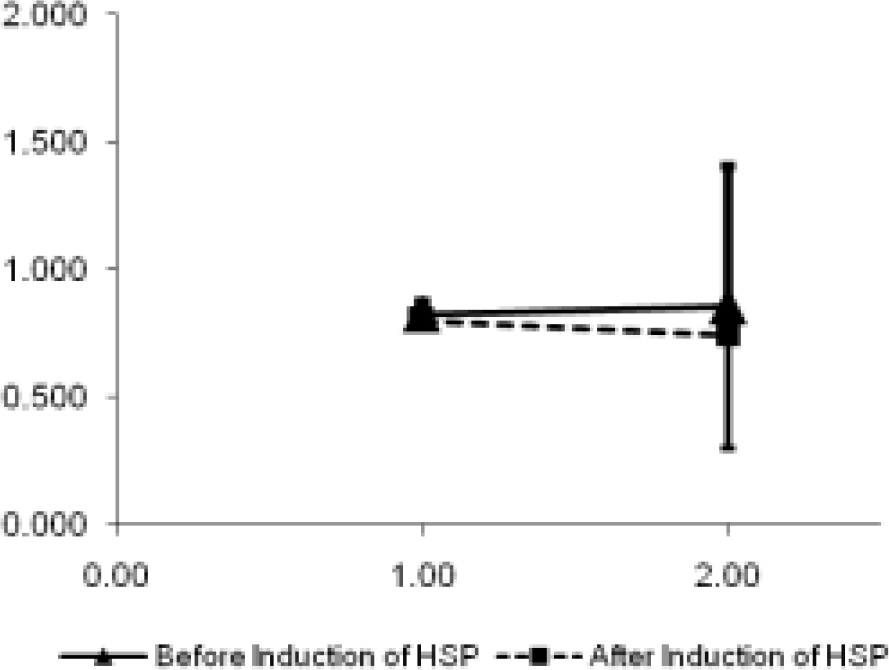
- Reaction time at ground and at 18,000 ft
Correct responses
It is observed from Table 3 that the significant main effect of HSP (F=29.862, p=0.000398) was observed on correct responses. Post hoc analysis for this response was carried out by LSD test and significance value are shown in Table 3.
The significant interaction effect of HSP x alt was observed on correct responses (F=9.656, p=0.0126). Post hoc analysis for individual comparison of this response is shown in Table 3 .
Reaction time
Significant main effect of HSP (F=32.710; p=0.000287) and significant interaction effect of HSP x Alt (F=44.237; p=0.000094) was observed on reaction time for responses. Post hoc analysis for individual comparison is shown in Table 3.
Comparison of correct responses and reaction time before and after induction of HSP at ground level and at 18,000 ft is shown in Table 4.
| Groups | Correct responses | Reaction time |
|---|---|---|
| A vs B | 0.023 | 0.0005 |
| A vs C | 0.030 | 0.002 |
| A vs D | 0.002 | 0.000015 |
| B vs C | 0.0005 | 0.000005 |
| B vs D | 0.00006 | 0.00000 |
A and B denote values at ground level and 18,000 ft before induction of HSP C and D denote values at ground level and 18,000 ft after induction of HSP
Discussion
The present study was conducted to examine the effect of induction of HSP-70 on performance on a working memory task in hypoxia. Working memory task was a computer based visual working memory test in the form of correct responses, incorrect responses and reaction time. The subjects were evaluated both before and after induction of heat shock proteins. The altitude of 18,000 ft was chosen as Armed Forces aviators frequently fly at these altitudes. Exposure duration at 18,000 ft was limited to 30 minutes as it was intended to evaluate the task performance of the subjects within the established time of useful consciousness (TUC), which is 25-30 minutes at 18,000 ft.
In the present study, the significant main effect of HSP (F=29.862, p=0.000398) was observed on correct responses. When the data for correct responses was pooled together for HSP by collapsing the data for altitude, it showed that the number of correct responses changes from 29.0 in absence of HSP to 30.6 in presence of HSP. The significant interaction effect of HSP x Alt was observed on correct responses (F=9.656, p=0.0126). Whenever, a significant interaction effect of two independent variables on a dependent variable is observed, the significant main effect of either of the two variables is overlooked and not considered important. In the present study, the significant interaction effect of HSP and altitude on correct responses was observed. Hence, significant main effect of HSP was not considered meaningful. This implied that the induction of HSP significantly modified the response at two levels of altitude i.e. at ground level and at 18,000 ft.
In reaction time responses, significant main effect of HSP (F=32.710; p=0.000287) and significant interaction effect of HSP x alt (F=44.237; p=0.000094) was observed. The reaction time was observed to be reduced from 0.841 sec in absence of HSP to 0.786 sec in presence of HSP when the data was pooled together for induction of HSP. Like correct responses, the significant interaction effect of HSP and altitude on reaction time was considered meaningful in spite of having a significant main effect of HSP. This indicates that HSP induction attenuated the reaction time response in the altitude.
The role of HSP to act as a cyto-protective agent during stress response is reported by many investigators. Studies have reported that prior induction of HSPs by a mild stress has a protective effect against a more severe stress [10].
The mechanism of neural protection conferred by induced HSP is not clear. Most HSPs have strong cyto-protective effects and are involved in many regulatory pathways. They have attempted to give possible mechanism of action of cytoprotection. The popular theory amongst all is chaperone concept [11,12,13,14]. Chaperones are a functionally related group of proteins that assists in protein folding in correct manner in bacteria, plant and animal cells under physiological and stressful conditions. Folding of proteins in correct manner in the cell is important to prevent cell injury. Inappropriate folding leads to cellular damage. Chaperones, especially the stress inducible HSP-70, have been studied for their potential to protect the brain from ischemic injury [15]. An important subgroup of highly evolutionarily conserved chaperones is the ATP-dependent heat shock proteins (HSPs), which share the ability to recognize and bind nascent and unfolded proteins, thus preventing aggregation, and facilitating correct protein folding [16,17]. Thus, regulation of the state of protein folding and protein association is a central aspect of normal cellular homeostasis, which is severely perturbed by ischemia and reperfusion. Despite a large number of studies demonstrating neuroprotection by the chaperone HSP-70, in both animal stroke studies [18,19] and cell culture models of ischemia [20] the mechanism, or more likely mechanisms, of protection are poorly understood.
The improvement in correct responses and reaction time while performing tasks in hypoxia in the present study may be attributable to the HSP induction and its regulatory role in correct folding of the protein in the brain. In the working memory task the subject is required to identify the target stimulus in an array of 4 stimuli. Quick processing of the sensory input in the brain and efficient motor output is required to accomplish the task. The processing of the information in the brain might have become faster due to induction of HSP and its probable role in processing activities of the brain.
Conclusion
The present study examined the effect of induction of heat shock protein on correct responses and reaction time in a working memory task in hypobaric hypoxia at 18,000 ft. The significant interaction effect of heat shock protein and altitude was observed on the correct responses and reaction time in hypoxia. This indicated that induction of heat shock protein improved the performance score and reduced the reaction time at two levels of altitude i.e. at ground level and at 18,000 ft. The limitation of the present study was that the study was conducted only on 10 subjects. For a more valid and meaningful scientific result the future studies are recommended with large number of subjects. Other realistic operational aircrew performance task like Flight Oriented Performance Task (FOPT) may be employed to evaluate task performance in hypoxia following induction of heat shock protein for future studies.
Recommendation
Exposure to hypoxia impairs the working memory task as evident from the results of correct responses and reaction time in a visual memory task in the present study. Keeping the psychomotor performance intact during high altitude flying is very important for military aviation. There are numerous reports regarding improvement of neuronal functions due to induction of HSP. The induction of HSP-70 due to heat stress prior to hypoxic stress is able to confer protection in hypoxic challenge in the present study.
Conflict of interest: None
References
- The effect of moderate levels of simulated altitude on sustained cognitive performance. Int J of Av Psycho. 1999;9(4):351-9.
- [Google Scholar]
- Cognitive function at high altitude. Human Factors: The Journal of the human factors and ergonomics Society. 1993;35(2):329-44.
- [Google Scholar]
- Psychomotor skills learning under chronic hypoxia. Neuroreport. 1999;10(14):3093-9.
- [Google Scholar]
- The degree of protection provided to neuronal cells by a pre-conditioning stress correlates with the amount of heat shock protein 70 it induces and not with the similarity of the subsequent stress. Neuroscience letters. 1995;200(2):85-8.
- [Google Scholar]
- Ann Neurol. 2000;47:782-91.
- [Google Scholar]
- Neurosci Lett. 1997;224:9-12.
- [Google Scholar]
- Ann Neurol. 1998;44:584-91.
- [Google Scholar]
- Heat shock protein 70 and stimulation induced by +Gz. Space Med Med Eng (Beijing). 2001;14(6):460-4.
- [Google Scholar]
- Synthesis and degradation of heat shock proteins during development and decay of thermotolerance. Cancer Res. 1982;42:2457-61.
- [Google Scholar]
- Heat shock proteins and heat adaptation of the whole organism. J Appl Physiol. 1997;83:1413-7.
- [Google Scholar]
- Molecular chaperone function of mammalian HSP 70 and HSP 40–A review. Int J Hyperthermia. 2000;16:231-45.
- [Google Scholar]
- The neuroprotective potential of heat shock protein 70 (HSP70) Mol Med Today. 1999;5:525-31.
- [Google Scholar]
- Chaperons, protein aggregation and brain protection from hypoxic/ ischemic injury. J Exp Bio. 2004;207:3213-20.
- [Google Scholar]
- Folding of newly translated proteins in vivo: the role of molecular chaperones. Annu Rev Biochem. 2001;70:603-47.
- [Google Scholar]
- Transgenic mice expressing the human inducible Hsp70 have hippocampal neurons resistant to ischemic injury. Cell Stress Chaperones. 1997;2:162-7.
- [Google Scholar]
- Heat-induced protection of mice against thermal death. Cancer Res. 1983;43:5758-60.
- [Google Scholar]
- Over-expression of Hsp-70 protects astrocytes from combined oxygen-glucose deprivation. Neurorep. 1996;7:429-32.
- [Google Scholar]






